Mamta Manglani1, P Balamurugan2.
1Professor Pediatrics, Division of Pediatric Hematology-Oncology, LTMM College and LTMG Hospital, Sion, Mumbai,
23rd Year Resident Chief, Division of Hematology-Oncology Department of Pediatrics, In-charge,Pediatric HIV Clinic, Division of Pediatric Hematology-Oncology, LTMM College and LTMG Hospital, Sion, Mumbai. | When evaluating a child with a potential bleeding disorder, it is extremely crucial to ascertain whether the patient's symptoms are appropriate to a bleeding disorder and whether further investigation of an underlying bleeding disorder is required. To understand approach to a bleeding child, it is essential to know the steps in hemostasis.
Physiology of Hemostasis
Hemostasis is a delicate balance between clot formation, anticoagulation and fibrinolysis and it occurs in the following phases:
- Vascular response
- Platelet adhesion
- Platelet aggregation
- Clot formation
- Clot stabilization
- Limitation of clot by anticoagulation
- Fibrinolysis to re-establish vascular patency
The primary phase of hemostasis is the production of the platelet plug. This process begins immediately after vascular injury as subendothelial tissues are exposed to circulating blood. Platelet binding to subendothelial tissue i.e. adhesion is followed by platelet activation with release of platelet contents, leading to platelet aggregation. The platelet plug not only offers the first line of defense against hemorrhage but also provides the phospholipid surface required for coagulant protein's interactions necessary for the formation of a fibrin clot.
The secondary phase of hemostasis results in the formation of a fibrin clot. The coagulation proteins circulate in inactive precursor forms; however, with the initiation of coagulation, these are converted to active forms. These activated proteins then further activate other factors in chain reaction (Appendix 1), ultimately leading to the formation of the blood clot. Although the clotting cascade model has undergone significant modification since its introduction in 1964, it is still very useful for the interpretation of abnormal coagulation test results such as the prothrombin time (PT) and activated partial thromboplastin time (aPTT).
The interactions of activated platelets and the clotting cascade give rise to hemostatic response that is potentially explosive and unchecked, may lead to thrombosis and tissue damage. Fortunately, coagulation is modulated by a number of mechanisms including the removal of activated factors through the reticuloendothelial system and the control of activated procoagulants by natural antithrombotic pathways (antithrombin III, protein C, protein S). In addition to these limiting reactions, there exists the fibrinolytic pathway, which restores vessel patency following hemostasis.
History and Clinical Examination :
To begin with, it is important to address the following questions in any child who is referred for bleeding. These include:
- Is it bleeding?
- If it is bleeding, then is it localized or generalized?
- If generalized, is it platelet type or coagulation type of bleeding?
- Is it congenital/hereditary or acquired disorder?
- What are the factors affected?
History and physical findings should enable the pediatrician to identify patients with a high likelihood of having a bleeding disorder. First of all, one needs to confirm whether there is true bleeding or is it some other lesion confused with bleeding e.g. fixed drug eruption, erythema nodosum, which may appear like ecchymoses. Fixed drug eruption appears at the same site each time a particular drug is taken. It does not change its color over time. Erythema nodosum occurs over extensor surfaces and are generally tender and red. Viral exanthem and mosquito bites may be confused with petechial rash; however petechial rash does not blanch on slide test. If it is bleeding, one needs to know whether it is local or generalized bleeding? If there is only a single site involved, it is more likely to be a localized bleeding rather than a generalized bleeding disorder. If epistaxis is from a single nostril and repeatedly from the same site, it is highly likely that there is a local cause for bleeding. On the other hand, if epistaxis is occurring from both the nostrils with or without evidence of bleeding from other sites it is likely a generalized disorder. If it is a generalized bleeding consider a bleeding disorder, but address the question is it congenital or acquired? Features of abnormal bleeding include a longer duration than routine bleeding episodes and more severe than routine bleeding. When a child has had previous surgery or dental extractions without bleeding complications, it is unlikely there is an underlying congenital hemorrhagic disorder.
The time of onset of symptoms is important. Acute onset over a period of days to weeks is suggestive of an acquired disorder, such as immune thrombocytopenic purpura (ITP) or vitamin K deficiency. Symptoms of a longer duration are indicative of a congenital disorder such as von Willebrand Disease (vWD) or coagulation-factor deficiencies. In case of infants with congenital coagulation disorders, bleeding manifestations sometimes occur at birth (following circumcision), appear in the first months of life (with immunizations) or most commonly, present when children become mobile and begin to experience mild trauma. Mild bleeding disorders may not become apparent until a person experiences surgery, dental extractions, trauma, or menstruation. In some children, bleeding with trauma is associated with good initial hemostasis, followed by a delayed oozing. This type of bleeding characteristically is seen with factor XIII deficiency and disorders of the fibrinolytic pathway.
The type of bleeding manifestation helps guide the evaluation. Mucosal bleeding (gum bleeds, epistaxis, menorrhagia), petechiae, and bruising (Fig.1) are more typical of quantitative or qualitative platelet disorders and von Willebrand disease (vWD). Conversely, spontaneous deep muscle (Fig.2) and joint bleeding (Fig.3) is seen more commonly with bleeding due to coagulation-factor deficiencies such as in hemophilia.
Fig. 1: Petechiae & Ecchymoses

Family history of the patient is important in the diagnosis of a congenital bleeding disorder. Sometimes, inherited hemorrhagic disorders have gone undiagnosed in families or have been misdiagnosed for multiple generations, especially when mild. Questioning should include all members of the family, regardless of any early suspicions as to the nature of diagnosis. For example hemophilia, an X-linked disorder that primarily affects males may also result in abnormal bleeding symptoms in female carriers. It is important to inquire about previous surgical procedures, dental extractions and transfusions in all family members. The menstrual and obstetric histories of female relatives can throw light on certain bleeding disorders.
History of drug ingestion is crucial as results of tests may be misleading and wrong diagnosis may be made. Non-steroidal anti-inflammatory drugs such as ibuprofen, mefenamic acid, etc. and aspirin inhibit platelet function whereas anticonvulsants, antihistaminics, antituberculous drugs especially rifampicin are known to cause thrombocytopenia.
Fig 2 - Deep muscle bleed

Fig 3: Joint Bleed (Hemarthrosis)

The overall health of the patient also is a clue to the cause of bleeding. Congenital bleeding disorders and ITP usually occur in children who are otherwise well. Conversely, disseminated intravascular coagulation (DIC) occurs in sick individuals with co-morbid conditions. Liver disease often is associated with bleeding due to impaired production of coagulation factors and renal disease is often associated with abnormalities in platelet function, thereby leading to defects in primary hemostasis.
Physical examination offers further clues to the diagnosis. Petechiae are pathognomonic of platelet-related bleeding, and mucus membrane bleeds (Fig.4) may also be present. Ecchymoses in places other than the anterior shins and elbows, such as the flank, abdomen, or back, may be indicative of a bleeding propensity. Bruises in any area that appear excessively large for the degree of trauma or those with underlying palpable hematomas may be seen in patients with significant bleeding disorders. Swelling of any joint without a history of significant trauma is definitely abnormal. Similarly, deep tissue and intramuscular bleeds should prompt the diagnosis of a coagulation factor deficiency. However, the possibility of physical abuse must be considered in the evaluation of any child with unusual patterns of bruising or bleeding.
Fig 4: Mucous membrane bleed - subconjunctival, periorbital & gum bleed

Laboratory investigations in a bleeding disorder:
The initial laboratory studies to evaluate a suspected bleeding disorder should include a complete blood count (CBC) with platelet counts as well as a review of the smear, especially for clumping of platelets and confirmation of the platelet count obtained on the counter. In addition, tests such as Prothrombin time (PT), an activated Partial thromboplastin time (aPTT), thrombin time, plasma fibrinogen levels and factor XIII assay are important in arriving at a specific diagnosis. Results of these tests will determine subsequent studies to be obtained.
Complete Blood count :
The complete blood count offers at least two important pieces of information. It allows for rapid determination of the platelet count, either confirming or rejecting a suspected thrombocytopenia. Anemia in association with a history of bleeding symptoms could suggest prolonged blood loss. A microcytic anemia, indicative of iron deficiency, may represent a history of prolonged blood loss, not compensated for by normal dietary iron intake. Alternatively, a normocytic anemia may be seen in cases of recent hemorrhage, blood loss. On the other hand, a normocytic or macrocytic anemia may suggest a hemolytic anemia e.g. in Evan's syndrome (Autoimmune Hemolytic Anemia & ITP). Apart from anemia, if leucopenia is also present along with thrombocytopenia, it raises the suspicion of bone marrow failure syndromes as seen in aplastic anemia, leukemia and lymphoma.
Peripheral blood smear :
Peripheral blood smear should be examined in every patient with a suspected platelet disorder. Examination of the smear allow for corroboration of platelet counts obtained on an automated cell counter as well as looking at platelet morphology. In platelet functional disorders such as Glanzmann's thrombasthenia, Bernard-Soulier Syndrome, platelets will be seen isolated with no clumps on smear (confirm non EDTA smear) In the giant platelet disorders such as Bernard-Soulier or May- Hegglin anomaly, the majority of platelets will be of a size similar to or larger than the erythrocytes. On a smear from a patient with the much more common ITP, both normal and large platelets are seen. Conversely, the Wiskott-Aldrich syndrome is characterized by smaller-than-normal platelet volume. The mean platelet volume is often reported as part of an automated complete blood count but may not accurately reflect the actual platelet size, particularly in the presence of thrombocytopenia.
PT and aPTT :
The PT and aPTT are screening tests for the second phase of hemostasis. PT evaluates the extrinsic (factor VII) and common pathway (factor X, V and II) of the coagulation cascade, whereas the aPTT evaluates the intrinsic (factors VIII, vWF, IX, XI and XII) and common pathways. PT is often reported as an international normalized ratio (INR), a standard allowing for the comparison of results between different laboratories. Prolongation of the PT or aPTT is indicative of a factor deficiency. The factor level at which either the PT or aPTT becomes prolonged varies but is usually around 40% of normal pooled plasma levels for any given factor.
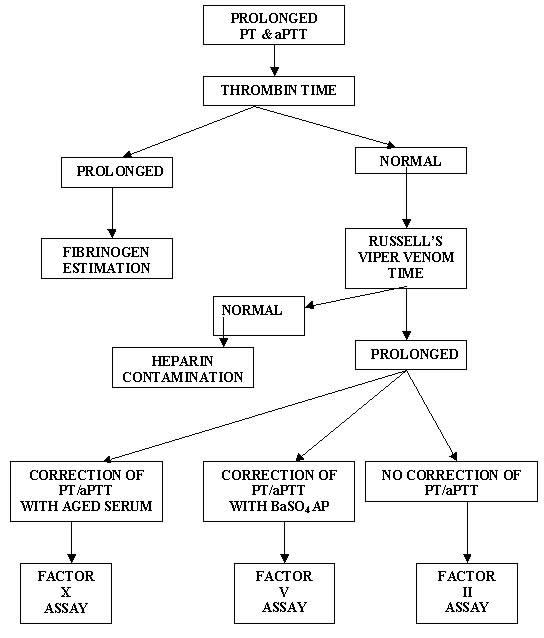
APTT and PT mixing studies :
An abnormal PT or aPTT should be followed by a mixing study, the results of which will indicate either the presence of an inhibitor or a factor deficiency. By mixing equal volumes of a patient's plasma with normal plasma, any factor deficiency should be corrected to a minimum of 50% levels; hence, the normalization of the PT or aPTT following a mixing study indicates a factor deficiency. Persistent prolongation of the test result after mixing with normal plasma is indicative of the presence of an inhibitor (usually an antibody against one or more coagulation factors). If BaSO4 adsorbed plasma is added to patient's plasma, and the aPTT normalizes, it indicates factor VIII deficiency, if aged serum added to patient's plasma normalizes the aPTT, it indicates factor IX deficiency. If it normalizes with both, it is suggestive of factor XI deficiency. When aPTT mixing studies indicate a factor deficiency, it is necessary to measure factors VIII, IX and XI because deficiencies of these factors are associated with clinical bleeding. Decreased concentration of factor XII, prekallikrein, and high molecular weight kininogen also can cause a prolongation of the aPTT; however these deficiencies are not associated with bleeding. If there is a prolongation of aPTT and platelets are not in clumps on the peripheral smear, it strongly suggests vWF deficiency.
Thrombin time (TT) :
A prolonged TT signifies low fibrinogen activity (hypofibrinogenemia or dysfibrinogenemia), the presence of fibrin spilt products, or heparin contamination. The reptilase clotting time is similar to the thrombin time, except that the coagulation induced by this enzyme from snake venom is not inhibited by heparin. For this reason, utilizing TT and reptilase clotting time together can help clarify whether abnormal coagulation results are due to heparin contamination.
Appendix 2 shows the interpretation of various coagulation tests & Appendix 3 & 4 depict the mixing studies.
Screening tests for defects in primary Hemostasis :
In addition to the coagulation-factor tests described above, it often is important to screen for abnormalities in primary hemostasis such as vWD or platelet-function abnormalities. The traditional test used has been the template bleeding time and often is included as part of initial screening for a bleeding disorder. Questions regarding the reliability, sensitivity, specificity, and predictability of the bleeding time, however, have led to a decline in its use. Platelet aggregation studies are extremely useful in identifying an abnormality in platelet function. It should be noted that normal numbers of platelets are needed for primary hemostasis and no functional tests should be done in patients with thrombocytopenia.
Platelet Aggregation studies :
Platelet aggregation studies measure the degree and pattern of platelet aggregation after the addition of platelet agonists (adenosine diphosphate, epinephrine, collagen, thrombin, arachidonate, ristocetin). The patterns of aggregation observed can aid in the detection and definition of platelet function disorders such as storage pool defects, Bernard-Soulier syndrome, or Glanzmann's thrombasthenia. Platelet aggregation testing and interpretation is typically performed only in specialized laboratories. Flow cytometry is a newer tool, which can be used to directly detect platelet membrane glycoproteins.
Clinical scenarios encountered in patients being evaluated for bleeding disorders The presentation of the bleeding patient can be quite variable and all instances are not addressed in this article. The common hemorrhagic problems associated with leukemia and aplastic anemia are not discussed here, except to mention that bruising and petechiae frequently are present at diagnosis and are often the reason the child is first brought to medical attention. The presence of anemia, leukocyte abnormalities, adenopathy, and/or hepatosplenomegaly, however, signifies distinct differences from other bleeding disorders and immediately changes the focus of inquiry.
- Significant bleeding history, normal PT, normal aPTT, and normal Platelet count :
The important consideration here is the clinician's impression that the bleeding history is significant. That being the case, despite the normal screening test results, further work-up is warranted. This scenario could be due to vWD, factor XIII deficiency, defects in fibrinolysis, or platelet function disorders.
- History of bleeding and isolated prolongation of the aPTT :
An isolated prolongation of the aPTT can be due to an inhibitor, heparin contamination, or deficiency in the intrinsic pathway of coagulation, excluding those factors in the common pathway (factors V, X, prothrombin, and fibrinogen). Of the remaining factors (high molecular weight kininogen, factors VIII, IX, XI, and XII), only deficiencies of three (VIII, IX, and XI) result in a bleeding diathesis. As discussed previously, vWD can present with this scenario, depending on the magnitude of factor VIII deficiency. Bleeding history with an isolated PT prolongation Factor VII is the only factor tested for by the PT that is not part of the common pathway. In the absence of an inhibitor, isolated prolongation of the PT is consistent with factor VII deficiency; however, factor VII deficiency is a rare entity and there are other possibilities that the clinician should consider. Because factor VII has the shortest half-life of all clotting factors, plasma levels drop most rapidly in situations affecting the production of multiple procoagulants (e.g., warfarin ingestion or acute liver failure). Thus, prolongation of the PT would be expected acutely, prior to prolongation of the aPTT. Similarly, in most laboratories, the PT is more sensitive to deficiencies of the vitamin K-dependent factors (factors II, VII, IX and X) than is the aPTT.
- History of bleeding and prolongation of both PT and aPTT
with a normal platelet count Prolongation of both the PT and aPTT in a symptomatic patient can be indicative of deficiency of one or more common pathway factors. Deficiency of a single factor in the common pathway (factor V, X, prothrombin) can prolong both of the coagulation times: however, such conditions are rare. Deficiency of multiple factors from both the intrinsic and extrinsic pathways may have a single etiology and also prolong both the PT and aPTT.
- History of bleeding and prolongation of PT and aPTT and thrombocytopenia :
This is most commonly encountered in sick children, with the bleeding and hematologic abnormalities reflecting dic or liver failure. A careful history and physical examination typically provides the diagnosis. Although the diagnosis is mainly clinical, laboratory testing can be used to support the diagnosis of dic. Typical laboratory abnormalities include decreased fibrinogen concentration, decreased factor V and VIII activity, and reduced levels of antithrombin III. Fibrin degradation products and D-dimers, which are specific for the degradation of fibrin, usually are elevated.
Liver is the primary site of synthesis for the majority of procoagulant and fibrinolytic proteins and protease inhibitors. Liver dysfunction due to any number of diseases results in an imbalance in the hemostatic system, often resulting in predisposition toward bleeding in response to trauma or surgical procedures. As in dic, both the PT and aPTT are typically prolonged due to deficiencies in multiple factors. In contrast to dic, factor VIII levels are typically normal or elevated. Thrombocytopenia may also be present due to portal hypertension and associated splenic sequestration.
- A child with petechiae and/or mucosal bleeding associated with isolated thrombocytopenia :
ITP in children most commonly is acute, but 10-15% of children have chronic ITP. Acute ITP is a self-limited acquired bleeding disorder due to the production of autoantibodies targeted against the patient's platelets. The typical presentation is an otherwise healthy child, with an acute onset of bleeding symptoms; most often diffuse petechiae and bruising. Less commonly, epistaxis, oral bleeding, hematuria, and gastrointestinal bleeding may be seen. Besides thrombocytopenia, there are no other hematologic abnormalities, although mild anemia may be present if there has been significant hemorrhage. Review of the smear sometimes reveals typical lymphocytes due to recent viral illnesses, but there are no other leukocyte abnormalities. Both large and normal sized platelets are seen.
- Asymptomatic child with a prolonged aPTT or PT :
This scenario is usually encountered when a child has screening coagulation studies obtained prior to a surgical procedure such as a tonsillectomy. Although the utility of such screening is debatable, the abnormal studies must be explained. The causes of prolonged PT and aPTT associated with bleeding have already been discussed; however, in the child without previous bleeding history and a negative family history, the approach to diagnosis may take a different path. The most common cause of such abnormal results is a circulating inhibitor, ultimately not associated with bleeding. Other causes of abnormal coagulation screening tests without a true bleeding disorder include deficiency of the contact factors prekallikrein, high molecular weight kininogen, or factor XII. Deficiencies of these factors may prolong the aPTT, but do not cause bleeding. On the contrary, factor XII deficiency is thought to represent a mild risk factor for thrombosis.
Conclusion
Children are likely to experience bruising and bleeding as normal events. The pediatrician must be able to distinguish such normal events from defective hemostasis. A thorough medical history and physical examination should enable the clinician to identify those children who deserve to undergo further evaluation. With this approach in mind, pediatricians should be capable of performing the initial laboratory evaluation and making the correct diagnosis in majority of cases.
Appendix 1: PHYSIOLOGY OF HEMOSTASIS

Appendix 2: INTERPRETATION OF TESTS OF HEMOSTASIS

Appendix 3 : MIXING STUDIES

Appendix 4 : MIXING STUDIES
 | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Thomas AE. The Bleeding Child; Is it NAI? Arch Dis Child 2004; 89 : 1163 - 1167. [CrossRef] [PMC free article]
- Hampton KK, Preston FE. ABC of Clinical Hematology : Bleeding Disorders, Thrombosis and Anticoagulation. BMJ 1997; 314 : 1026 - 1032. [CrossRef]
- Allen GA, Glader B. Approach to the Bleeding Child. Pediatr Clin N Am. 2002; 49 : 1239 - 1256. [CrossRef]
- Lusher JM. Clinical and Laboratory Approach to the Patient with Bleeding. In Nathan and Oski's Hematology of Infancy and Childhood. 6th Edn. Eds. Nathan DG, Orki SH, Ginsburg D, Look AT. WB Saunders Company. 2003; pps 1515 - 1526.
- Montgomery RR, Scott JP. The Clinical & Laboratory Evaluation. In Nelson Textbook of Pediatrics. 17th Edn. Eds. Behrman RE, Kliegman RM, Jenson HB.WB Saunders Company. 2004; pps 1653 - 1656.
|
| Cite this article as: | | Manglani M, Balamurugan P. APPROACH TO A BLEEDING CHILD. Pediatr Oncall J. 2005;2. |
|